How Do You Know if Something Is a Good Nucleophile
Nucleophile
- Page ID
- 1148
4 Factors in Affecting SN ii Reactions
- The nature of the leaving grouping (SouthwardN2 Reactions-The Leaving Group)
- The reactivity of the nucleophile (SNii Reactions-The Nucleophile)
- The solvent (SNtwo Reactions-The Nucleophile)
- The structure of the alkyl portion of the substrate (Due southNii Reactions-The Substrate)
The Reactivity of the Nucleophile
At present that we have adamant what will brand a good leaving grouping, we volition now consider nucleophilicity. That is, the relative strength of the nucleophile. Nucleophilicity depends on many factors, including accuse, basicity, solvent, polarizability, and the nature of the substituents.
Increasing the Negative Charge Increases Nucleophilicity
Nucleophiles can be neutral or negatively charged. In either case, information technology is important that the nucleophile be a good Lewis base of operations, meaning it has electrons it wants to share. The following diagram is just a reminder of some of the nucleophiles that were presented in the department covering nucleophilic substitution. In looking at these two types of nucleophiles, you should notice that a reactive atom, such as oxygen, in a neutral species can also be a reactive atom in a negatively charged species. For example, the O in OH- is negatively charged, just the O in H2O is neutral.

It has been experimentally shown that a nucleophile containing a negatively charged reactive atom is better than a nucleophile containing a reactive atom that is neutral. The next diagram illustrates this concept. Notice that when oxygen is part of the hydroxide ion, information technology bears a negative charge, and when it is role of a water molecule, it is neutral. The O of -OH is a better nucleophile than the O of H2O, and results in a faster reaction rate. Similarly, when nitrogen is role of NH2, it bears a negative charge, and when it is function of NH3, information technology is neutral. The Due north of NH2 is a meliorate nucleophile than the N of NH3, and results in a faster reaction charge per unit.
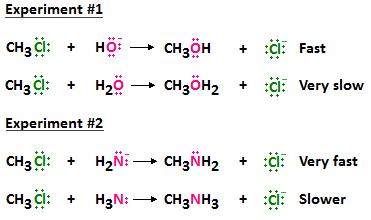
When Moving Beyond a Row, Nucleophilicity Follows basicity
To say that nucleophilicity follows basicity across a row means that, equally basicity increases from correct to left on the periodic table, nucleophilicity too increases. As basicity decreases from left to right on the periodic table, nucleophilicity too decreases. When it comes to nucleophilicity, do non assign this same rule when making comparisons betwixt the halogens located in a column. In this case of moving upwardly and downwardly a cavalcade, nucleophilicity does not always follow basicity. Information technology depends on the blazon of solvent you lot are using.

In the section Nucleophilic Substitution, we assigned a relationship to leaving groups containing C, N, O, and F, showing that the strength of the leaving group follows electronegativity. This is based on the fact that the best leaving groups are those that are weak bases that do non want to share their electrons. The best nucleophiles however, are proficient bases that want to share their electrons with the electrophilic carbon. The relationship shown below, therefore, is the exact contrary of that shown for the strength of a leaving group.
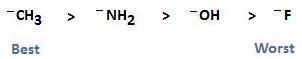
Solvents and Nucleophilicity
In full general chemical science, we classified solvents equally beingness either polar or nonpolar. Polar solvents tin exist further subdivided into protic and and aprotic solvents.
Protic Solvents
A protic solvent is a solvent that has a hydrogen atom bound to an oxygen or nitrogen. A few examples of protic solvents include HtwoO, ROH, RNH2, and R2NH, where h2o is an instance of an inorganic protic solvent, and alcohols and amides are examples of organic solvents. The diagram below shows a few examples of protic solvents we volition see.

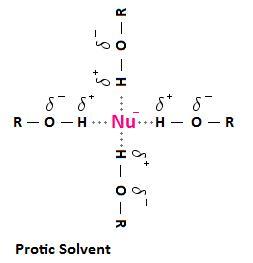
Since oxygen and nitrogen are highly electronegative atoms, the O-H and Northward-H bonds that are present in protic solvents result in a hydrogen that is positively polarized. When protic solvents are used in nucleophilic substitution reactions, the positively polarized hydrogen of the solvent molecule tin collaborate with the negatively charged nucleophile. In solution, molecules or ions that are surrounded past these solvent molecules are said to be solvated. Solvation is the process of allure and association of solvent molecules with ions of a solute. The solute, in this instance, is a negatively charged nucleophile.
The post-obit diagram depicts the interaction that tin can occur between a protic solvent and a negatively charged nucleophile. The interactions are called hydrogen bonds. A hydrogen bond results from a from a dipole-dipole force between between an electronegative atom, such as a halogen, and a hydrogen atom bonded to nitrogen, oxygen or fluorine. In the case below, nosotros are using an alcohol (ROH) as an example of a protic solvent, but be enlightened that this interaction tin can occur with other solvents containing a positively polarized hydrogen atom, such equally a molecule of water, or amides of the class RNHtwo and RiiNH.
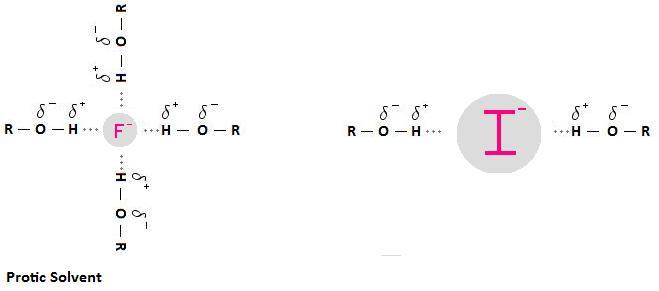
Why is this of import? Solvation weakens the nucleophile; that is, solvation decreases nucleophilicity. This is because the solvent forms a "shell" around the nucleophile, impeding the nucleophile's ability to attack an electrophilic carbon. Furthermore, because the charge on smaller anions is more concentrated, pocket-sized anions are more than tightly solvated than big anions.
The picture below illustrates this concept. Notice how the smaller fluoride anion is represented as beingness more heavily solvated than the larger iodide anion. This means that the fluoride anion will be a weaker nucleophile than the iodide anion. In fact, information technology is important to note that fluoride volition not function every bit a nucleophile at all in protic solvents. Information technology is and then pocket-sized that solvation creates a state of affairs whereby fluoride's lone pair of electrons are no longer accessible, meaning it is unable to participate in a nucleophilic substitution reaction.
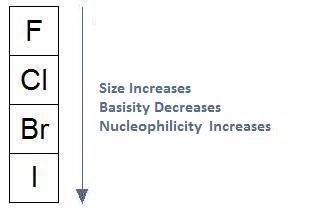
Previously we learned how nucleophilicity follows basicity when moving beyond a row. In our discussion on the effect of protic solvents on nucleophilicity, we learned that solvation weakens the nucleophile, having the greatest consequence on smaller anions. In effect, when using protic solvents, nucleophilicity does not follow basicity when moving up and down a column. In fact, it's the exact opposite: when basicity decreases, nucleophilicity increases and when basicity increases, nucleophilicity decreases.
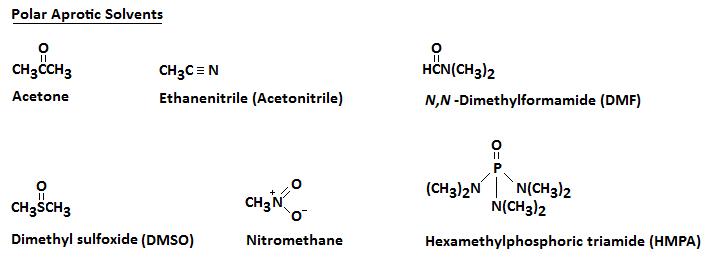
Aprotic Solvents
An aprotic solvent is a solvent that lacks a positively polarized hydrogen. The next diagram illustrates several polar aprotic solvents that you should become familiar with.
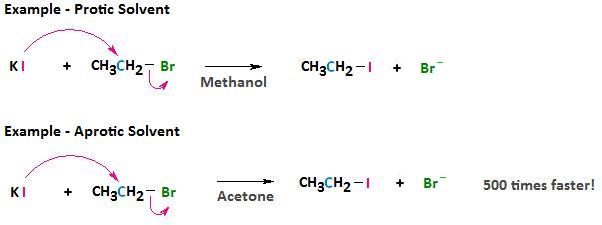
Aprotic solvents, similar protic solvents, are polar simply, because they lack a positively polarized hydrogen, they do not course hydrogen bonds with the anionic nucleophile. The result, with respect to solvation, is a relatively weak interaction between the aprotic solvent and the nucleophile.
The consequence of this weakened interaction is two-fold. First, past using an aprotic solvent nosotros can raise the reactivity of the nucleophile. This tin can sometimes have dramatic furnishings on the rate at which a nucleophilic substitution reaction can occur. For example, if nosotros consider the reaction betwixt bromoethane and potassium iodide, the reaction occurs 500 times faster in acetone than in methanol.
A second upshot that results from the weak interaction that occurs between aprotic solvents and nucleophiles is that, nether some conditions, there tin can exist an inversion of the reactivity order. An inversion would outcome in nucleophilicity following basicity up and down a column, as shown in the following diagram. When nosotros considered the furnishings of protic solvents, call up that the iodide anion was the strongest nucleophile. At present, in considering aprotic solvents nether some conditions, the fluoride anion is the strongest nucelophile.
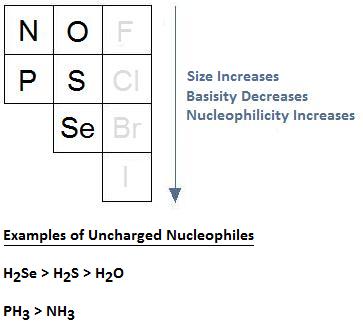
Increasing Atomic Size Increases Nucleophilicity
Thus far, our discussion on nucleophilicity and solvent selection has been limited to negatively charged nucleophiles, such as F-, Cl-, Br-, and I-. With respect to these anions we learned that, when using protic solvents, nucleophilicity does not follow basicity, and when using aprotic solvents, the same relationship can occur, or there could be an inversion in the order of reactivity.
What happens as we movement up and down a column when because uncharged nucleophiles? It turns out that, in the example of uncharged nucleophiles, size dictates nucleophilicity. This is considering larger elements have bigger, more than diffuse, and more polarizable electron clouds. This cloud facilitates the formation of a more effective orbital overlap in the transition country of bimolecular nucleophilic commutation (SN2) reactions, resulting in a transition country that is lower in free energy and a nucleophilic exchange that occurs at a faster rate.
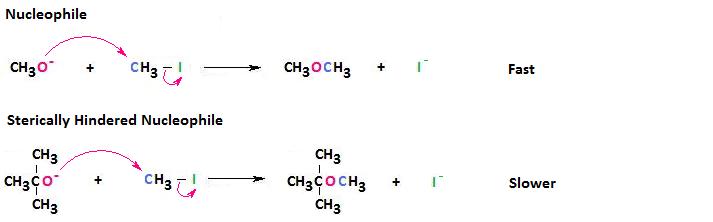
Sterically Hindered Nucleophiles React More Slowly
In the department Kinetics of Nucleophilic Substitution Reactions, we learned that the SN2 transition country is very crowded. Remember that there are a total of five groups around the electrophilic centre.
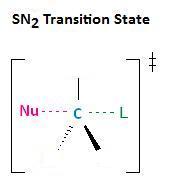
For this reason, sterically hindered nucleophiles react more slowly than those defective steric bulk.
Next section: SouthwardN2 Reactions-The Substrate
Contributors
- Rachael Curtis (UCD)
Source: https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Supplemental_Modules_(Organic_Chemistry)/Reactions/Substitution_Reactions/SN2/Nucleophile
0 Response to "How Do You Know if Something Is a Good Nucleophile"
Post a Comment